 
This area of the website celebrates the work of many famous scientists whose quest to learn more about the world we live in and the atoms that make up the things around us led to the periodic table as we know it today.Ĭan France claim the first periodic table? Probably not, but a French Geology Professor made a significant advance towards it, even though at the time few people were aware of it.Īlexandre Béguyer de Chancourtois was a geologist, but this was at a time when scientists specialised much less than they do today. It was not until a more accurate list of the atomic mass of the elements became available at a conference in Karlsruhe, Germany in 1860 that real progress was made towards the discovery of the modern periodic table. In 1829, Johann Döbereiner recognised triads of elements with chemically similar properties, such as lithium, sodium and potassium, and showed that the properties of the middle element could be predicted from the properties of the other two. Several other attempts were made to group elements together over the coming decades. The earliest attempt to classify the elements was in 1789, when Antoine Lavoisier grouped the elements based on their properties into gases, non-metals, metals and earths. Certainly Mendeleev was the first to publish a version of the table that we would recognise today, but does he deserve all the credit?Ī number of other chemists before Mendeleev were investigating patterns in the properties of the elements that were known at the time. (3) It's a lot easier to remember facts about 18 elements than over 100 elements.Ask most chemists who discovered the periodic table and you will almost certainly get the answer Dmitri Mendeleev. (2) These eighteen elements make up most of the matter in the Universe. Remember that the orbitals are the places you will generally find the electrons as they spin around the nucleus. (1) Electrons fit nicely into three orbitals. Since the launch of the site, we've been asked, "Why start with 18?" The rules for the first eighteen elements are very straightforward: You will never stop discovering new reactions and compounds, but the elements will be the same. With the tools you learn here, you can explore and understand the Universe. The iron atoms in the red soil of Mars are also the same. Iron (Fe) atoms found on Earth are identical to iron atoms found on meteorites. While there are more elements to discover, the basic elements remain the same. Scientists just confirmed the creation of element 117 in 2014. 
Up to this point in time, we have discovered or created about 120. Chemists have learned that over 95% of your body is made up of hydrogen (H), carbon (C), nitrogen, oxygen, phosphorus (P), and calcium (Ca).Īs far as we know, there are a limited number of basic elements. For example, you are made up of billions of billions of atoms but you probably won't find more than 40 elements (types of atoms) in your body. The term 'element' is used to describe atoms with specific characteristics. Remember that 'atom' is the general term. 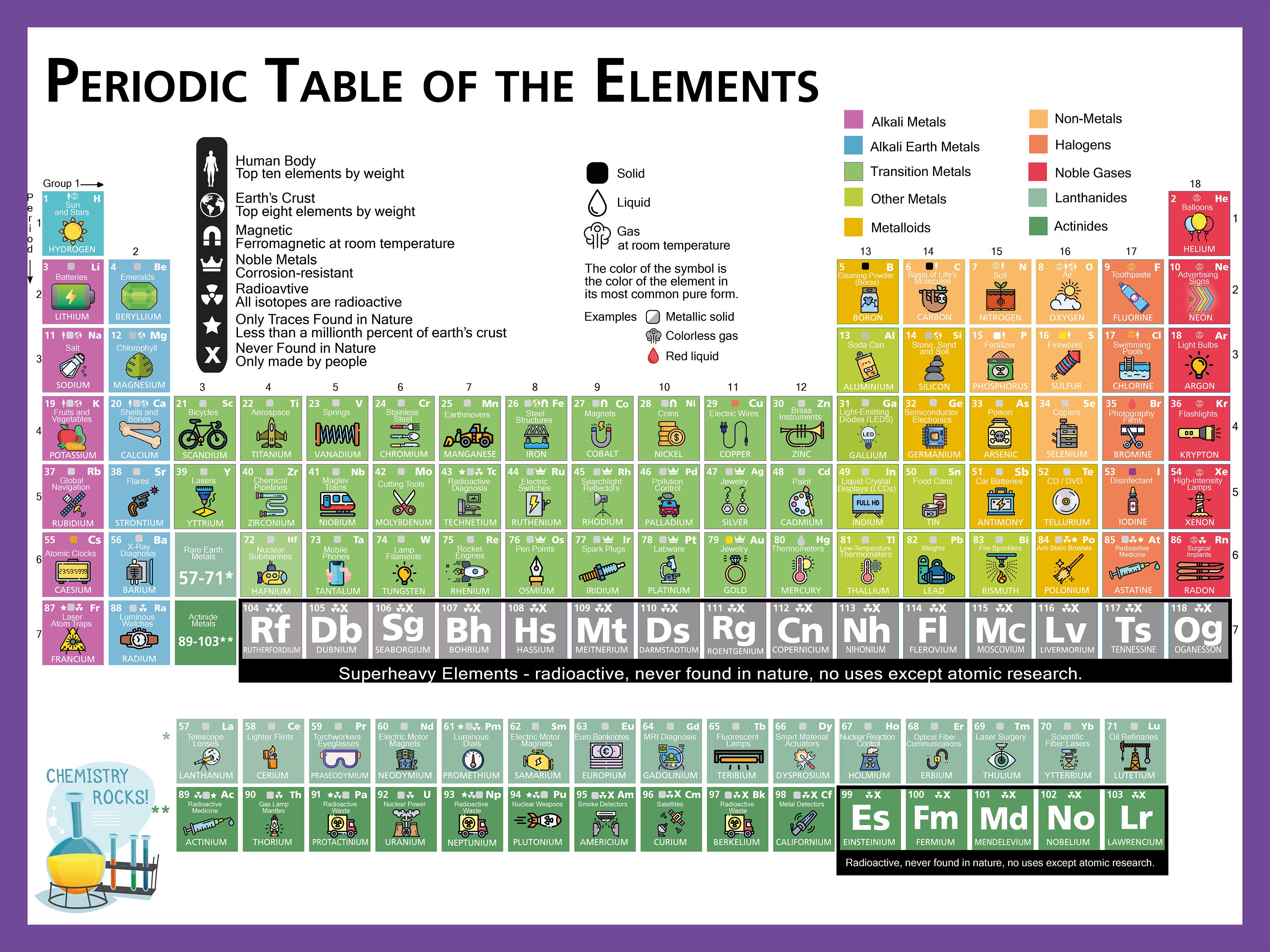
The atoms for each element are unique, even though they are all made of similar subatomic parts. If you have seven protons, neutrons, and electrons, you will have a nitrogen (N) atom. If you have eight protons, neutrons and electrons, you will have an oxygen (O) atom. When those pieces start combining in specific numbers, you can build atoms with recognizable traits. You know that a generic atom has some protons and neutrons in the nucleus and some electrons zipping around in orbitals. 
Now we're getting to the heart and soul of the way the Universe works. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
